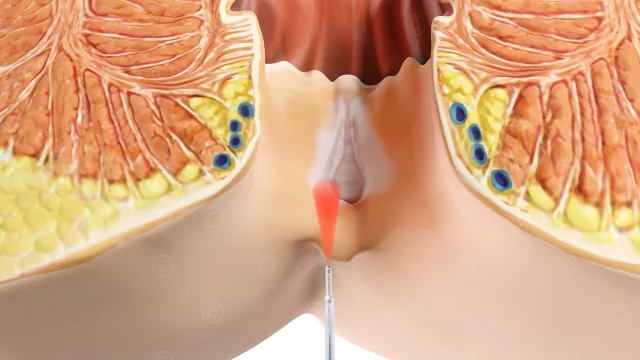
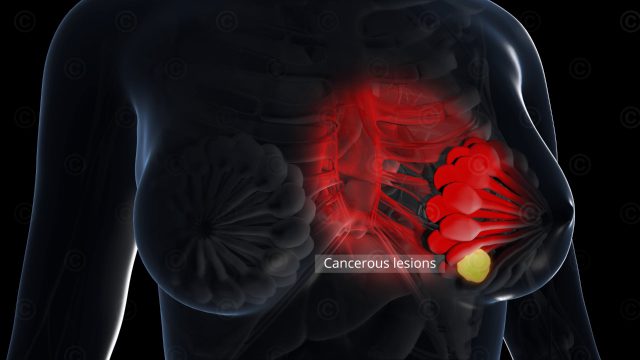
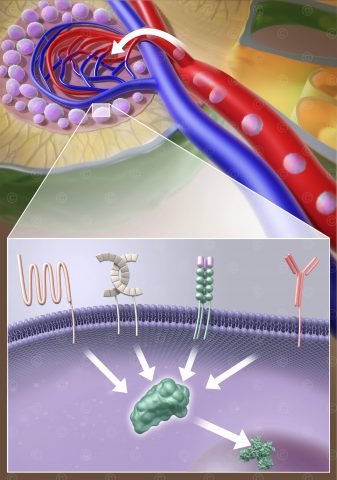
In the field of cancer research great hopes lay on new developments in cancer immunotherapy. The international biotechnology company Prima BioMed based in Sydney and Berlin is engaged in the development of drugs to strengthen the immune response in cancer. Prima BioMed requierd imagery for its medicine IMP321, whose approval is currently being tested in clinical trials.
Usage rights : Prima BioMed / Immutep
Music: Kevin McLeod – As I figure – License: Creative Commons (CC BY 3.0)
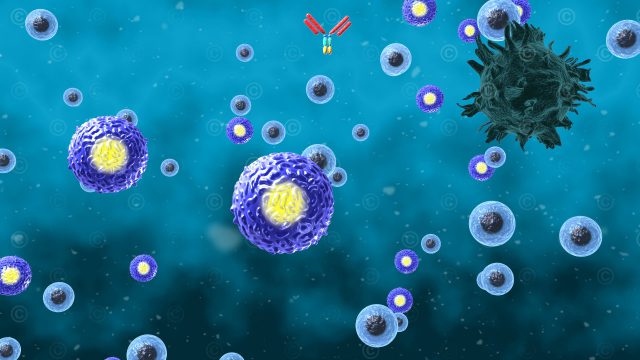
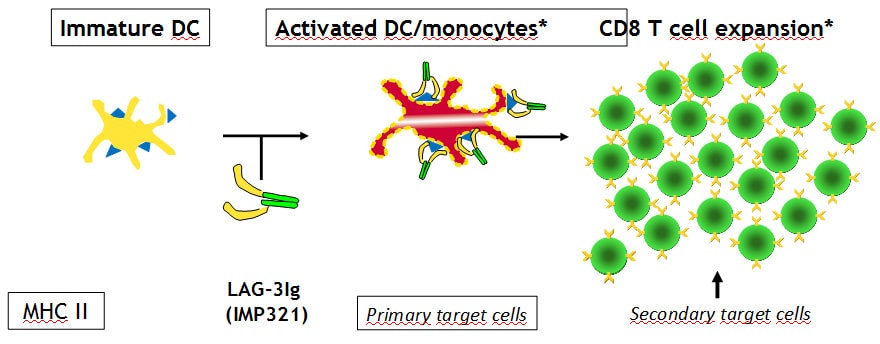
IMP321 is a soluble variant of the LAG3 immune checkpoint inhibitor. It enhances the immune response to cancer cells through the activation of antigen-presenting cells (dendritic cells) with simultaneous blockade of negative regulation by T cells. In the current study, the efficacy of IMP321 in metastatic breast cancer is investigated.
Illustrations:
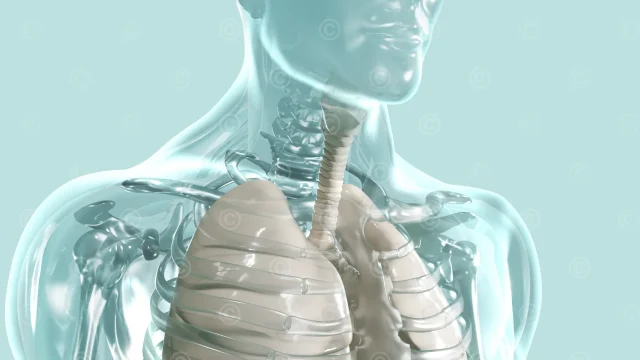
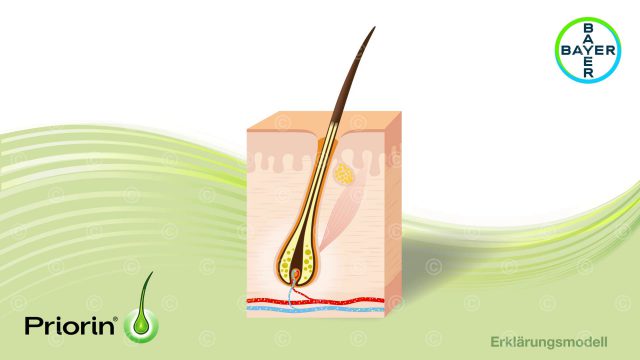
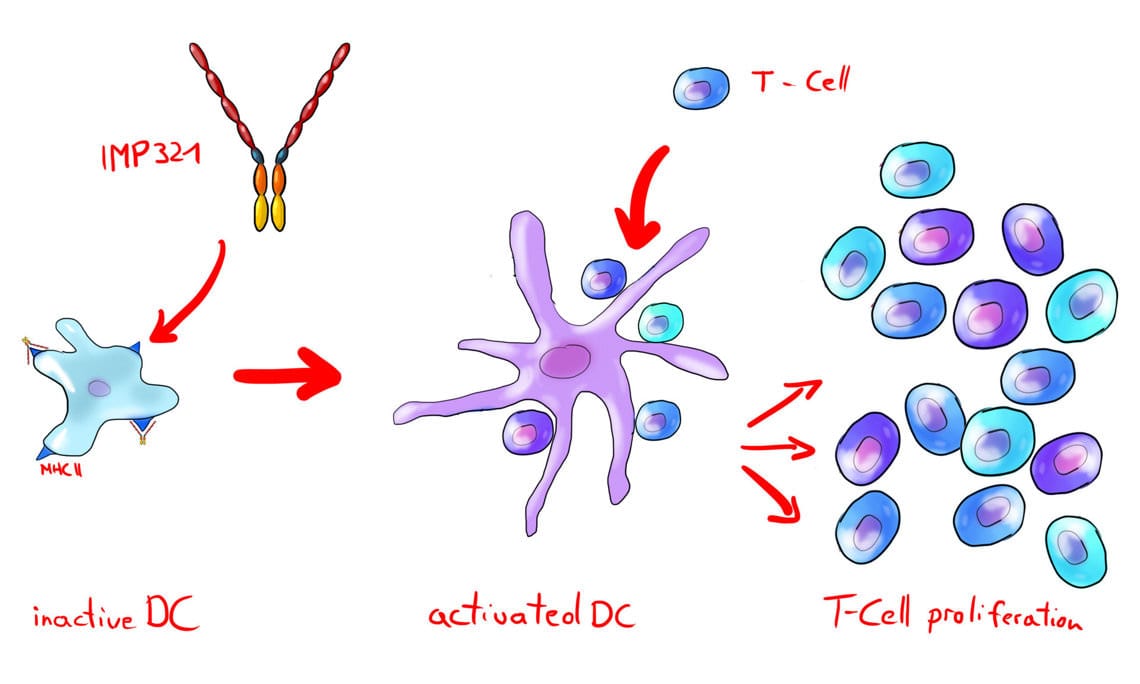
In the first step two illustrations of the mode of action and the technology behind it were created. Rough sketches, previously used in teachings, could be used for the definition of the contents.
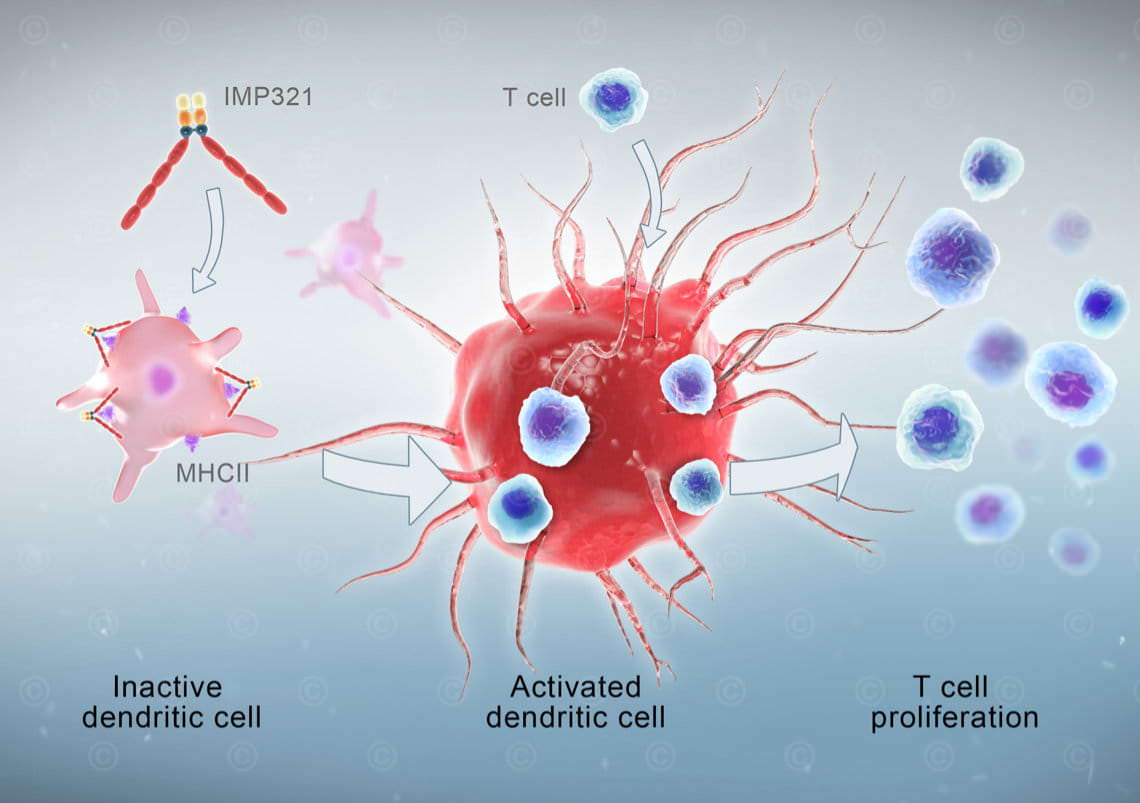
Through communication of various images, the desired style of the illustration could be established at an early stage. In the following step a sketch for each illustration was created and further substantiated in the subsequent process. The sketch-phase was very helpful to rule out misunderstandings or incorrect content weighting due to the complex subject and the immuno-biological communicate in english.
The desired look of the illustration, waxy and glassy structures, required a series of more complex shaders in calculating the final illustration. To achieve the desired appearance, refraction shaders and also sub-surface-scattering shader were used.
Animation MOA IMP321:
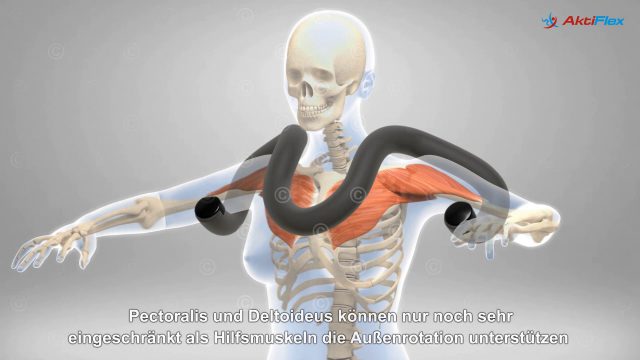
Based on the two previous illustrations a mode-of-action animation was created showing the mode-of-action of IMP321 in strengthening the immune response. This 3D animation was divided into three sections: the normal physiological situation with activation of dendritic cells by LAG3 proteins on T cells, the visualization of monoclonal antibodies as immune checkpoint inhibitors and compared to the previous the mechanisms of IMP321 in it´s process of strengthening the immune response.
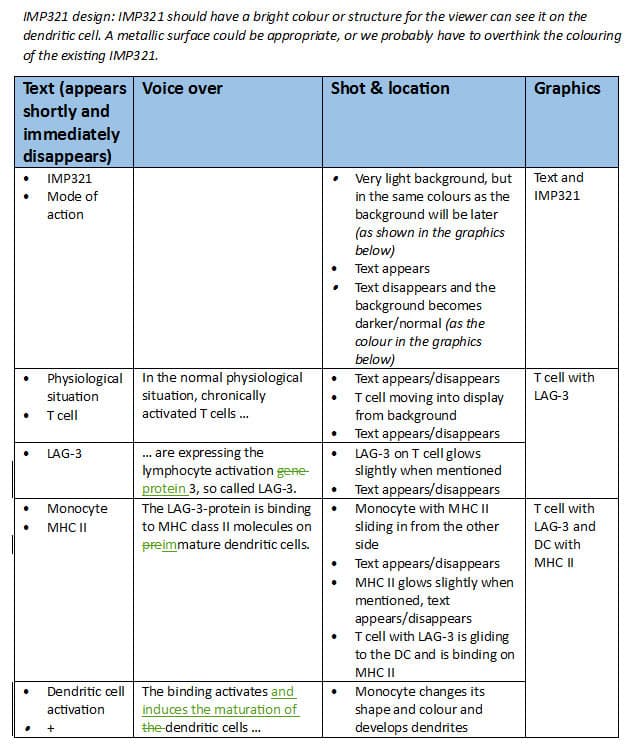
Prima BioMed delivered an already excellent and elaborate and well thought script to define the sequences of animation. On this basis a detailed storyboards were created and all parts of the film were clearly defined and coordinated with Prima BioMed.
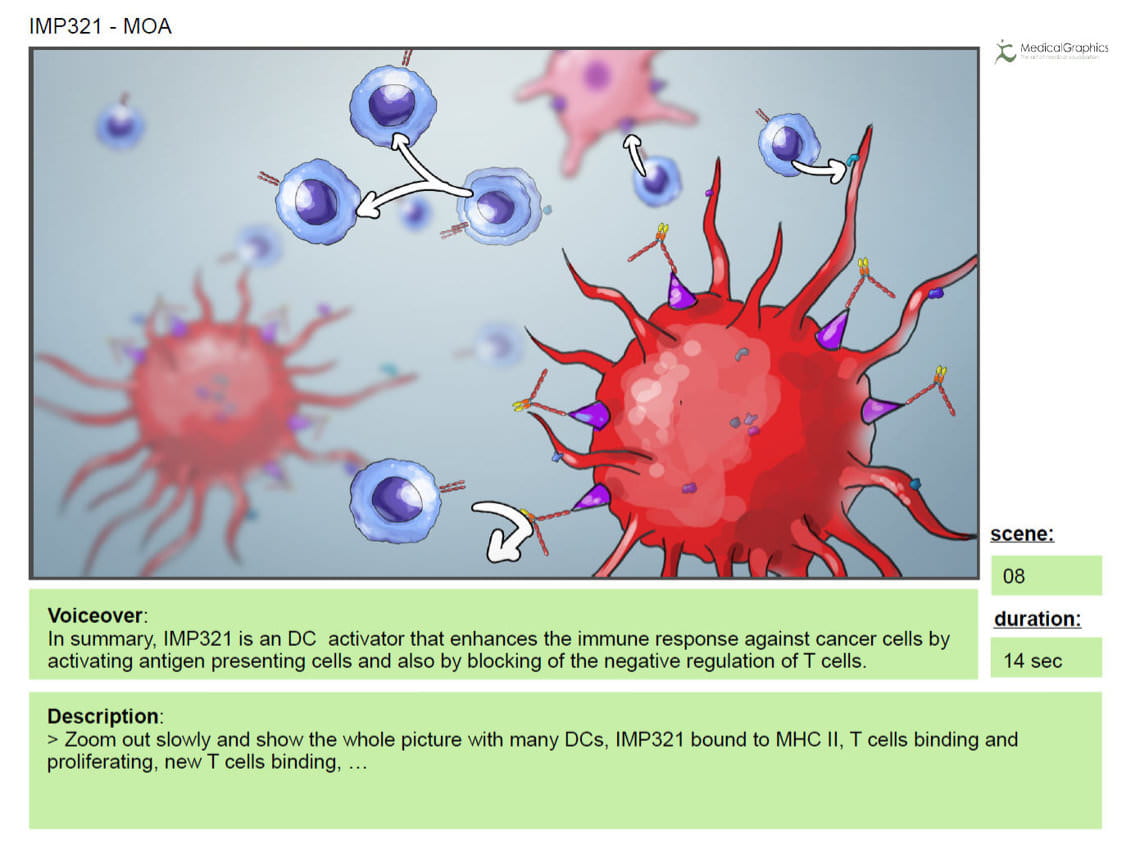
In the following Preview-phase the final voice recordings of the animation were ready to use. This allowed to adjust the timing of all sequences to the voice over in order to achieve the highest possible congruence of picture and content. Due to the depth of the content some inconsistencies within the storyboard occured while creating the (layout-) preview-animation. Since at this stage of a 3D animation project changes still can be done quite simply, the necessary adjustments could be made very fast.
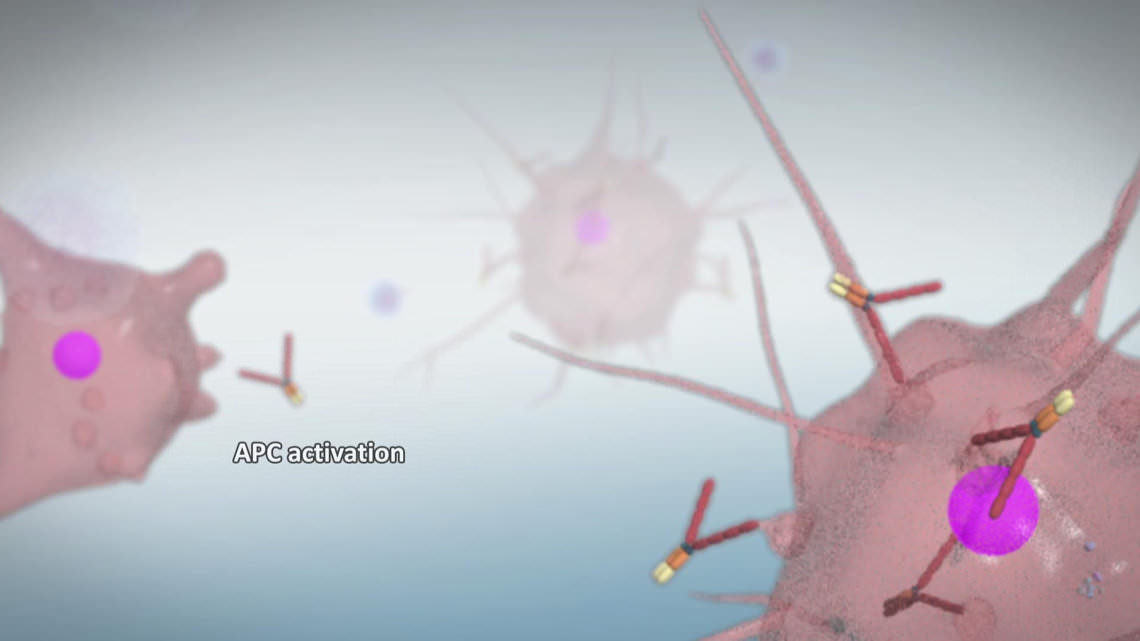
For the entire production only a very narrow time frame was available. Prima BioMed had a fixed date for the use of the film in a few weeks. To meet the deadline in the final production phase (calculating frames, assembling of the elements in compositing), some modifications to the objects and shaders were needed. In particular, the render times of the dendritic cells were to high, due to the SSS shader used.
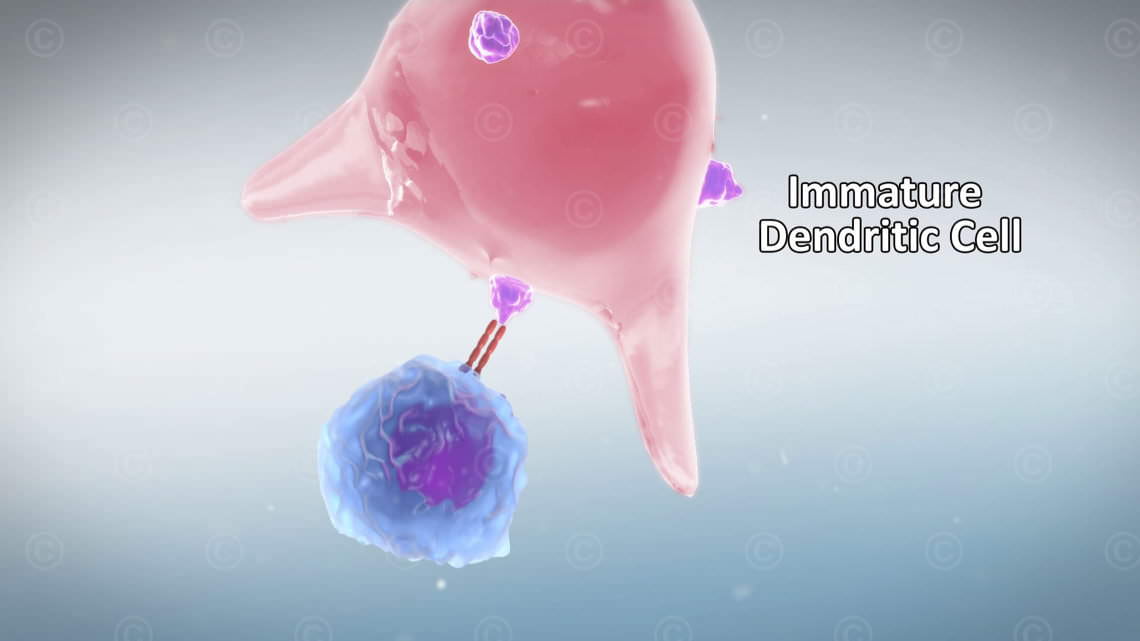
For the creation of the illustrations this was not significant, but for the animation, with its ~ 5000 frames, optimizations were here but necessary. We succeeded in reducing the rendering times by ¼, whereby a timely delivery of the animation nothing stood in the way.
The rights of the pictures and animations lie with “Prima BioMed”. The state of knowledge shown is as of December 2016. The active substance is in clinical trials and has not yet been approved.